|
Obtain further evaluation (e.g., ECG, echocardiogram) as indicated for patients who develop symptoms of arrhythmia (e.g., palpitations, lightheadedness, syncope, chest pain), new onset dyspnea, or other cardiovascular concerns. These events have occurred particularly in patients with cardiac risk factors including hypertension and diabetes mellitus, a previous history of cardiac arrhythmias, and in patients with acute infections.Įvaluate cardiac history and function at baseline, and monitor patients for cardiac arrhythmias and cardiac function. Grade 3 or greater ventricular tachyarrhythmias were reported in 0.2%, Grade 3 or greater atrial fibrillation and atrial flutter were reported in 3.7%, and Grade 3 or greater cardiac failure was reported in 1.3% of 4,896 patients who received IMBRUVICA ® in clinical trials, including in patients who received IMBRUVICA ® in unapproved monotherapy or combination regimens. Patients with cardiac comorbidities may be at greater risk of these events. These adverse reactions occurred in patients with and without preexisting hypertension or cardiac comorbidities. Deaths due to cardiac causes or sudden deaths occurred in 1% of 4,896 patients who received IMBRUVICA ® in clinical trials, including in patients who received IMBRUVICA ® in unapproved monotherapy or combination regimens. Monitor and evaluate patients for fever and infections and treat appropriately.Ĭardiac Arrhythmias, Cardiac Failure, and Sudden Death: Fatal and serious cardiac arrhythmias and cardiac failure have occurred with IMBRUVICA ®. Consider prophylaxis according to standard of care in patients who are at increased risk for opportunistic infections. Cases of progressive multifocal leukoencephalopathy (PML) and Pneumocystis jirovecii pneumonia (PJP) have occurred in patients treated with IMBRUVICA ®. Grade 3 or greater infections occurred in 21% of 1,476 patients with B-cell malignancies who received IMBRUVICA ® in clinical trials. Infections: Fatal and non-fatal infections (including bacterial, viral, or fungal) have occurred with IMBRUVICA ® therapy. Monitor for signs and symptoms of bleeding.Ĭonsider the benefit-risk of withholding IMBRUVICA ® for at least 3 to 7 days pre- and post-surgery depending upon the type of surgery and the risk of bleeding. Consider the risks and benefits of anticoagulant or antiplatelet therapy when co-administered with IMBRUVICA ®. The addition of antiplatelet therapy with or without anticoagulant therapy increased this percentage to 4.4%, and the addition of anticoagulant therapy with or without antiplatelet therapy increased this percentage to 6.1%. Across clinical trials, 3.1% of 2,838 patients who received IMBRUVICA ® without antiplatelet or anticoagulant therapy experienced major hemorrhage. 
Use of either anticoagulant or antiplatelet agents concomitantly with IMBRUVICA ® increases the risk of major hemorrhage. The mechanism for the bleeding events is not well understood. 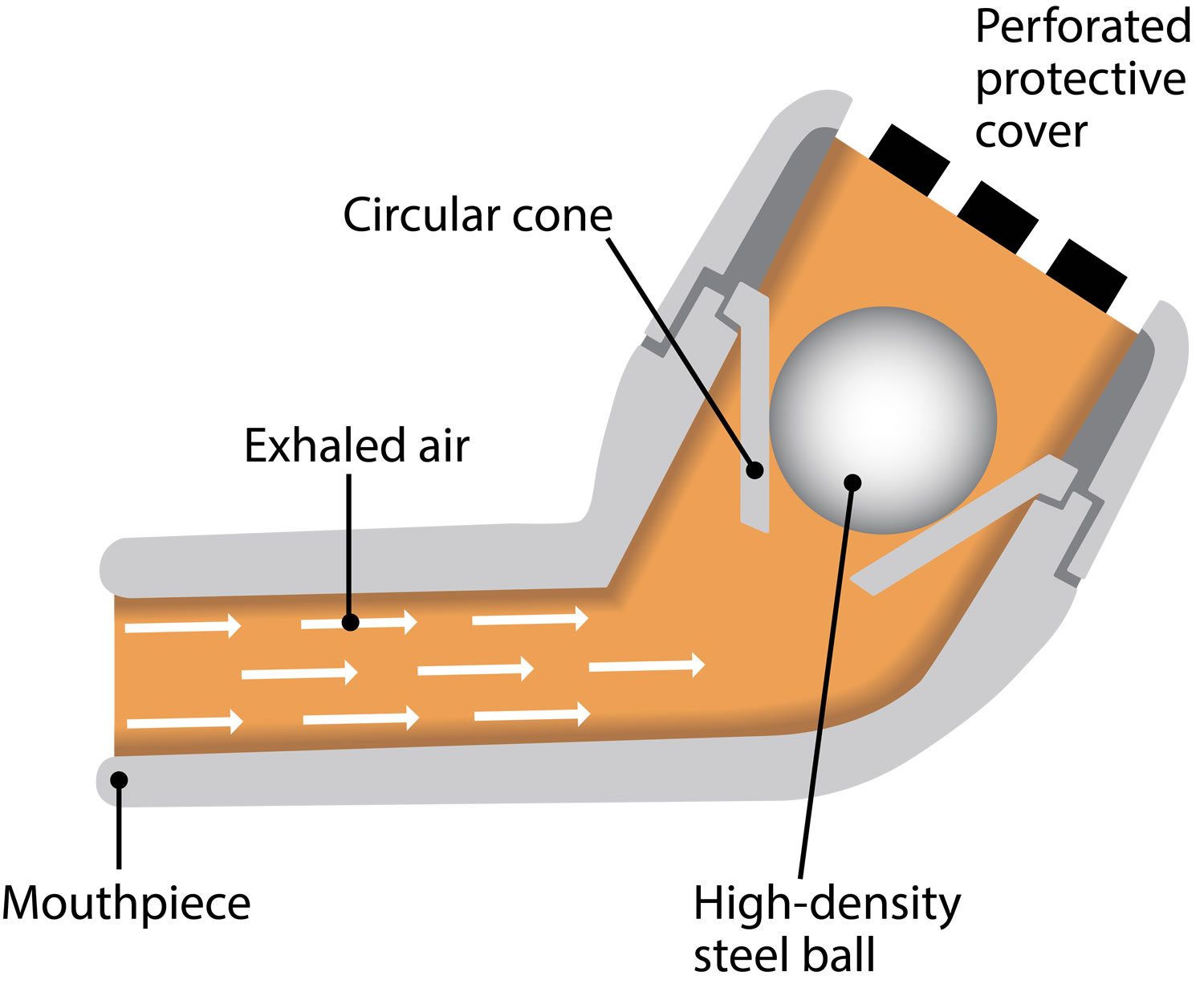
Bleeding events of any grade including bruising and petechiae occurred in 39%, and excluding bruising and petechiae occurred in 23% of patients who received IMBRUVICA ®, respectively. Major hemorrhage (≥ Grade 3, serious, or any central nervous system events e.g., intracranial hemorrhage, gastrointestinal bleeding, hematuria, and post procedural hemorrhage) occurred in 4.2% of patients, with fatalities occurring in 0.4% of 2,838 patients who received IMBRUVICA ® in 27 clinical trials. The null hypothesis was rejected and the primary effectiveness endpoint passed, since the lower bound of the one-sided 95% confidence interval was 94.95%, which was greater than the 90% performance goal.Hemorrhage: Fatal bleeding events have occurred in patients who received IMBRUVICA ®.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
